Skutterudites
| Skutterudites are built up from binary compounds with the chemical composition CoAs3. Binary CoAs3 was discovered
as a minaral on the farm Skutterud (Norway) in 1772 and served as the basis for the precious blue pigment used in luxury goods like fine china.
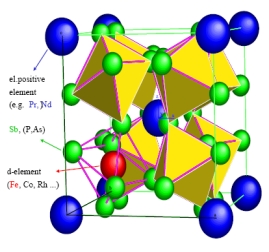
CoAs3 is a diamagnetic semiconductor characterised by a large void in its crystal structure. Co can be exchanged by Rh or Ir, while As
can be substituted by P and Sb without changing the crystalline unit cell. The cavities of skutterudites can be filled up by electropositive
elements like monovalent earth alkali (e.g., Na), divalent alkali (like Ba) or trivalent rare earth elements (La).
Since these electropositive elements are just loosely bound, heat carrying phonons are intensely scattered on the resulting „rattling modes“.
As a consequence, lattice thermal conductivity is dramatically reduced (by about one order of magnitude) driving a similar enhancement of
the thermoelectric figure of merit.
Our research activities in this field are focused on:
Collaborations: Uni Wien, CNRS Thias (Paris), MPI CPfS Dresden. |
Structure of skutterudite
|
Contact: E. Bauer, S. Bühler-Paschen, H. Michor |